JYMed Peptide’s New Semaglutide DMF No. 041090 Listed as “Available for Reference” by the U.S. FDA
JYMed Peptide recently announced that an additional Drug Master File (DMF) for its Semaglutide active pharmaceutical ingredient (API), DMF No. 041090, has successfully passed the U.S. Food and Drug Administration’s Completeness Assessment (CA) and is now listed as “Available for Reference.”
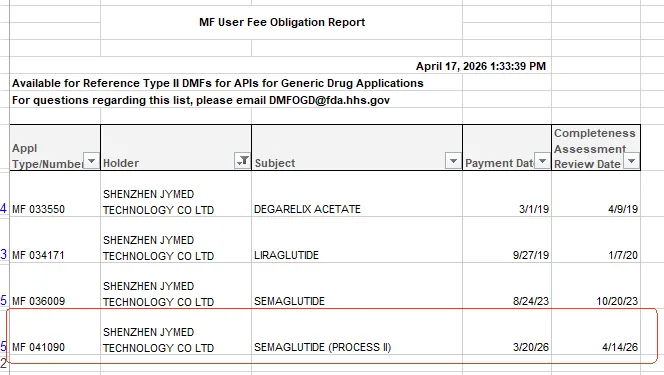
This new Semaglutide DMF Available for Reference further strengthens JYMed Peptide’s FDA compliance track record, following previous regulatory achievements for Degarelix Acetate, Liraglutide, and the company’s first Semaglutide DMF. Passing the Completeness Assessment confirms that the DMF meets the FDA’s foundational regulatory requirements, allowing formulation customers to reference it as a reliable compliance option for regulatory submissions. This helps lower the barrier for joint filings and accelerates global market collaboration.
With this milestone, JYMed Peptide now offers two referenceable Semaglutide DMFs, providing global formulation partners with greater flexibility, expanded API sourcing options, and stronger support for joint filings and international market development.
The listing of DMF No. 041090 reinforces JYMed Peptide’s competitive position in the rapidly growing GLP-1 therapeutic segment. It also reflects the company’s continued commitment to building a robust international regulatory platform and supporting customers in accelerating generic drug development.
Looking ahead, JYMed Peptide will continue expanding its global regulatory footprint, leveraging its growing portfolio of certifications and compliance capabilities to help partners streamline development, enter new markets, and achieve sustainable, high-quality growth.
About JYMed Peptide
JYMed Peptide is a high-tech enterprise specializing in the peptide sector, with business activities covering peptide API and formulation development, manufacturing, and sales; one-stop peptide CDMO services; cosmetic peptide ingredient development and manufacturing; and custom peptide synthesis services.
The company is committed to becoming a leading global provider of peptide solutions, delivering high-quality, compliant products and services to pharmaceutical and cosmetic customers worldwide.





0 Comments